The influence of anticoagulant EDTA-K2 and lithium heparin on PCT detection
Release time:
2020-11-18
Procalcitonin (PCT) is a calcitonin propeptide without hormone activity. It is a glycoprotein with a relative molecular mass of 13000 composed of 116 amino acids. Under physiological conditions, it is mainly produced by thyroid follicular cells, and its concentration is usually <0.1ng/mL. In recent years, PCT has played an important role in prompting bacterial infection and its degree of infection, judging the effect of treatment, and guiding antimicrobial treatment. effect.
As a clinical laboratory, the choice of anticoagulant for PCT detection is still a matter of concern. Therefore, Desheng Technology has carried out countless experiments and comparative discussions on this, mainly through the electrochemical immunoassay analysis of 13 cases The serum or plasma of the patients separated in the vacuum drying tube, separation hose, lithium heparin tube and EDTA-K2 tube were measured immediately, 12, 24, and 72 hours, and other experiments were compared and analyzed, and some conclusions were drawn.
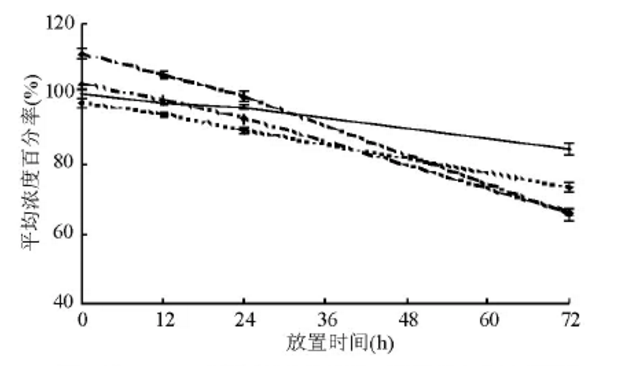
The average degradation percentage of serum and plasma of different anticoagulants at different storage time
1. The degradation rate of EDTA-K2 anticoagulant is slow
EDTA-K2 is the anticoagulant recommended by the kit, and the degradation rate of EDTA-K2 anticoagulated plasma PCT is the slowest within 72 hours. The average concentration of EDTA-K2 anticoagulated plasma immediately detected is used as the percentage standard reference. The average concentration detected by the drying tube is 102.84%, which has a difference of +2.84%; the separation hose is 111.47%, which has a difference of +11.47%, and the lithium heparin tube is 97.24%, which is -2. 76% difference.
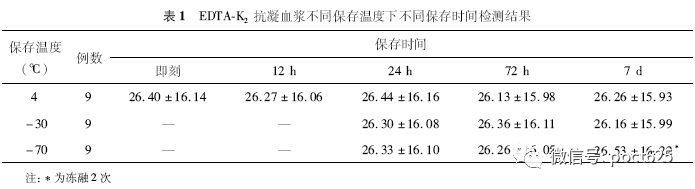
2. EDTA-K2 anticoagulated plasma test results for different storage times at different storage temperatures Even if the 9 EDTA-K2 anticoagulation specimens were stored at 4°C, there was no statistically significant difference from the immediate determination of the separated plasma at 7 days (P>0.05); they were stored at -70°C, and repeated freeze-thaw cycles were repeated twice for 7 days. The result has no obvious effect. See Table 1.
3. In the research of ordinary drying tube, separation hose, lithium heparin tube and EDTA-K2 tube, the test result of lithium heparin tube is lower than that of EDTA-K2 tube by 2.76%. It has been reported that the degree of heparin's influence on the labeled immunoassay varies with different methods and test items. This test uses electrochemiluminescence method for detection. It may be that heparin has strong acidity and negative charge, which will affect the catalytic environment of the chemical reaction, the buffer system and even the chemical reaction itself, making the detection result low.
In order to ensure the accuracy, timeliness and effectiveness of the test results, it is possible to reduce the impact of too long specimen placement time from improving the professional level of the inspectors, improving the work process, and shortening the test time. As an electrochemiluminescence method, this study It is confirmed that the use of EDTA-K2 as an anticoagulant to collect specimens not only shortens the specimen detection, but also has a good test result, and it has only a small degradation rate when stored for a long time. However, in order to ensure the accuracy of the results, each laboratory should use the anticoagulant recommended by its own method to conduct the test within the recommended time.
Contact details
Contact number
Address: C8, Guanggu United Science and Technology City, Ezhou City, Hubei Province
Fax:0711-3704 589
Follow us



